 Such categorization is set to become increasingly less relevant due to several reasons: the availability of effective, early treatment for leprosy has resulted in decreasing ocular complications unique to leprosy the longevity of patients has in turn increased the occurrence of age-related ocular complications among leprosy patients the integration of specialized leprosy services into the general health care system has altered the role of primary health care workers to evaluate any cause of decreasing vision rather than only ones that are directly related to leprosy and more often than not, especially in bacteriologically cured leprosy patients, distinguishing leprosy related ocular complications from those that are not directly associated with the disease is difficult. Chronic inflammatory demyelinating polyradiculoneuropathy with monoclonal gammopathy of undetermined significance affects a subgroup of patients with the neuropathy and a monoclonal gammopathy usually of IgG or IgA type.Ocular manifestations in leprosy have traditionally been categorized as those directly related to leprosy and those that are not directly related to the disease. Chronic inflammatory demyelinating polyradiculoneuropathy with severe axonal loss is a rare variant characterized by a relapsing-remitting course with evidence of axonal damage. Progressive chronic inflammatory demyelinating polyradiculoneuropathy is another variant, which shows a steadily worsening course without remission. Pure motor or pure sensory variants are very rare and may represent an intermediate stage of the classic disease. Diagnostic features include neurophysiologic evidence of demyelinating neuropathy, elevation of CSF protein level without pleocytosis, and neuropathologic findings of peripheral segmentary demyelination with perineural and endoneural inflammatory infiltrates. Many genes are involved in this syndrome, and cerebellar hypoplasia is a feature in many chromosomal disorders.Īcquired disorder of peripheral nerves, affecting motor and sensory fibers of the limbs, reaching the nadir of deficits 8 weeks or later from onset, and exhibiting a relapsing-remitting or progressive course. Dysgeneses of the forebrain, including most of the primary disorders of neuroblast migration, may be associated CNS findings. 
Prognosis depends on underlying associated diseases, especially if progressive. 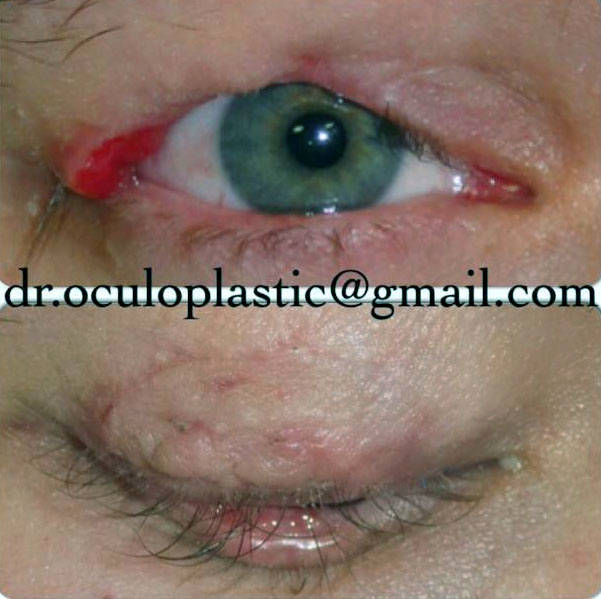
The basis pontis also may be hypoplastic. CT scan can often confirm the diagnosis, but MRI with sagittal images demonstrates it best. Cerebellar hypoplasia may result in histochemical changes in the muscle biopsy of fiber-type predominance or congenital muscle fiber-type disproportion, but necrotizing and dystrophic alterations are not found. The most constant clinical findings in infancy are gross motor developmental delay and generalized muscular hypotonia inconstant findings include nystagmus, corticospinal tract deficits, mental retardation, and epilepsy. Global cerebellar hypoplasia sometimes occurs without other cerebral dysgeneses. Vermal hypoplasia may leave a gap in the cerebellar midline, as with the autosomal recessive Joubert syndrome, or may be absence of the vermis with fusion of the medial surfaces of the cerebellar hemispheres and dentate nuclei, a condition known as rhombencephalosynapsis. It may be global or, less commonly, involves only the vermis selective involvement of the lateral cerebellar hemisphere with preservation of the vermis also is described but is rare. Ultimately the nitrogen is excreted, mostly as urea.Ī heterogeneous condition associated with other cerebral malformations, metabolic disorders, and degenerative diseases beginning in fetal life. The gut takes up glutamine released by these organs where it is an energy source. 
Ammonia that escapes the action of ammonia-utilizing enzymes in the liver or is released into the circulation from systemic metabolic processes is metabolized largely to glutamine in the extrahepatic organs: brain, skeletal muscles, heart, adipose tissue, and lungs. Gut-derived ammonia in the liver is converted largely to urea and to a lesser extent, glutamine. Even in the absence of bacteria in the intestine, ammonia is released by enzymatic hydrolysis of urea. Colon bacteria synthesize one-third to one-half of intestinal ammonia, and the rest comes from metabolic products and to some extent from the reabsorption of nutritional ammonia. 
Two-thirds of the ammonia comes from the gut from precursors of glutamine and urea. In catabolism muscular ammonia is in the foreground, and the purine nucleotide cycle in muscles and other tissues plays an important role. Ammonia is derived mainly from proteins, amino acids, amines, and nucleic acid, mainly in the gastrointestinal tract, kidneys, muscles, brain, liver, and red blood cells. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
